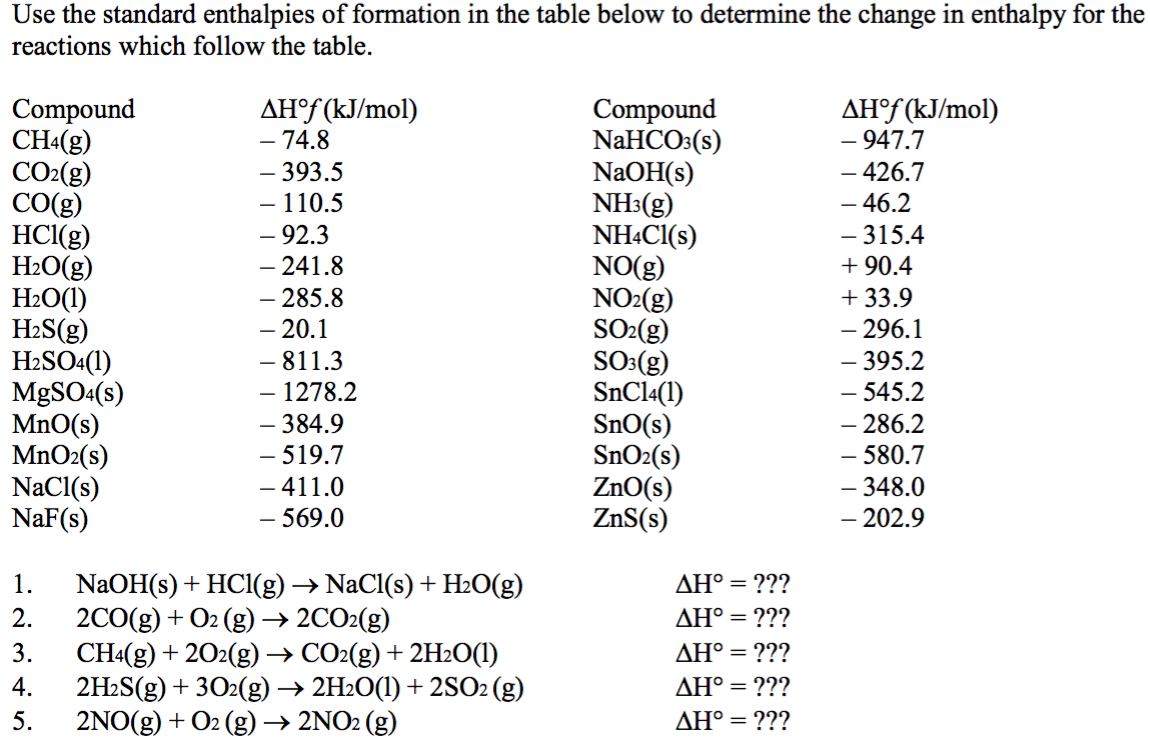
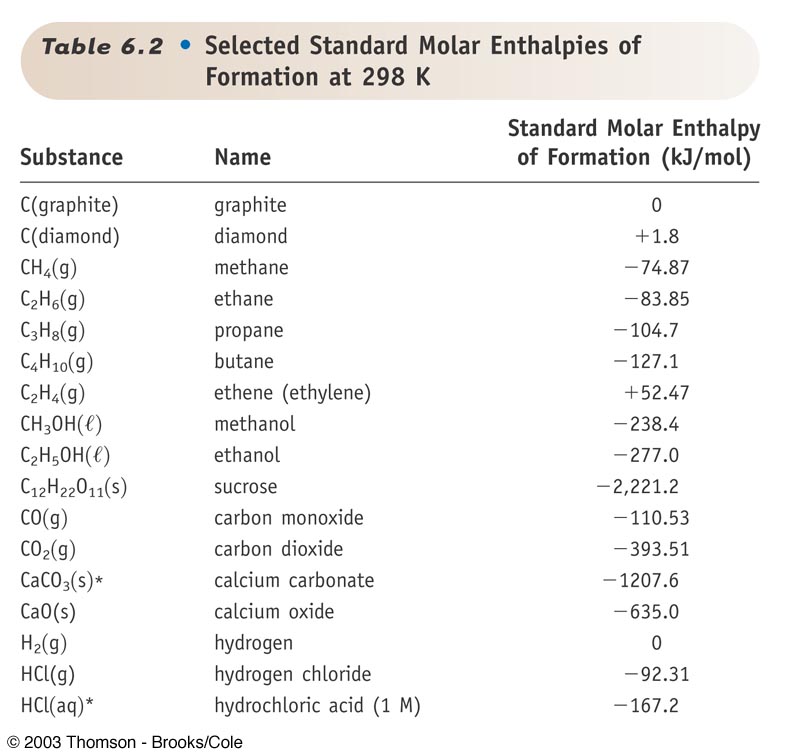
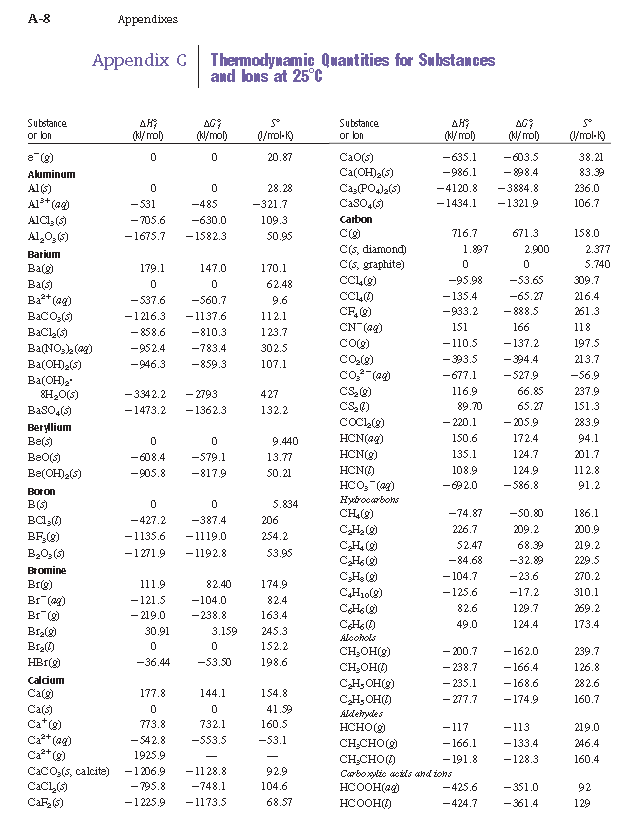
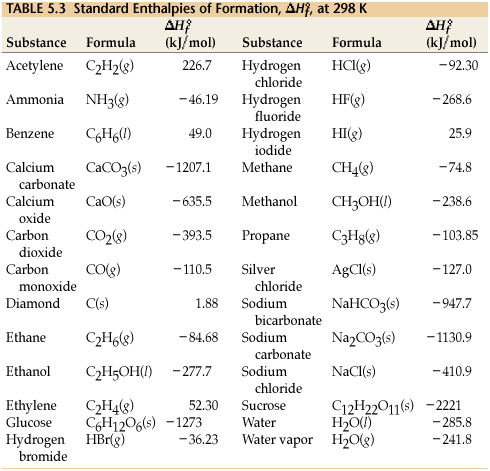
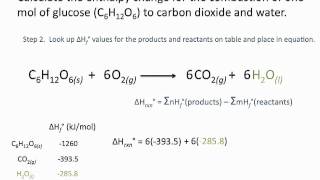![Appendix D. Heats of Formation and Combustion - Basic Principles and Calculations in Chemical Engineering [Book] Appendix D. Heats of Formation and Combustion - Basic Principles and Calculations in Chemical Engineering [Book]](https://www.oreilly.com/api/v2/epubs/9780132885478/files/graphics/appd-tab-d1a.jpg)
Appendix D. Heats of Formation and Combustion - Basic Principles and Calculations in Chemical Engineering [Book]

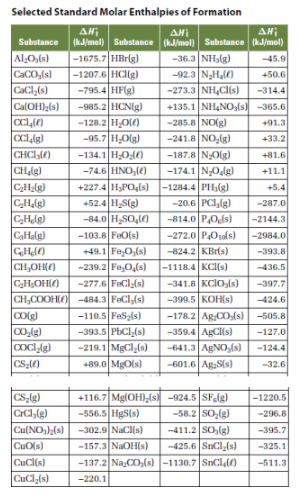
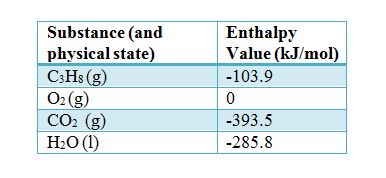
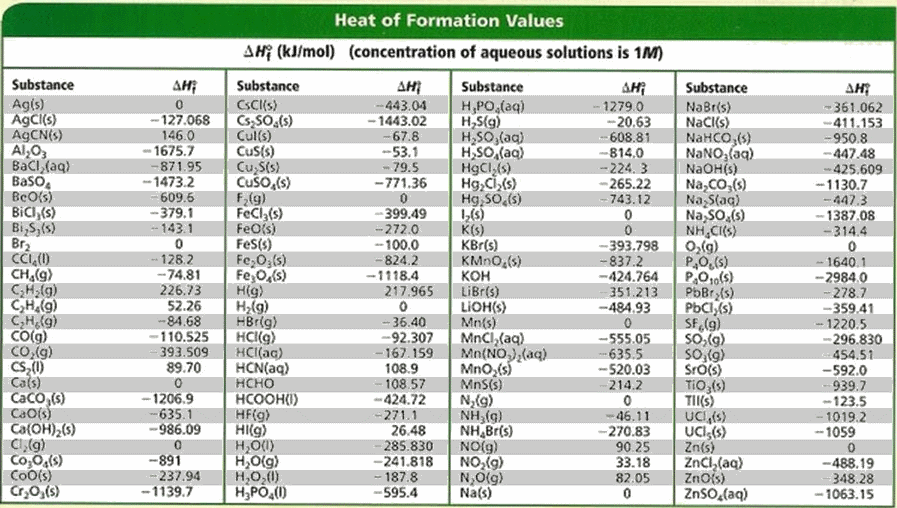
Table 6 from Structure and heats of formation of iodine fluorides and the respective closed-shell ions from CCSD(T) electronic structure calculations and reliable prediction of the steric activity of the free-valence electron

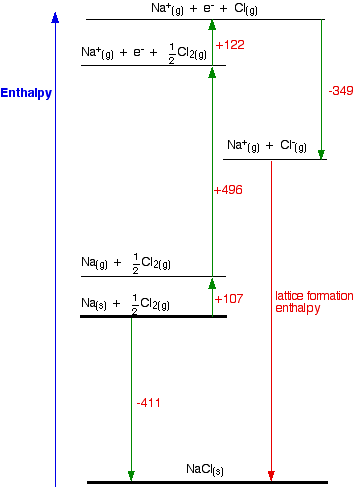
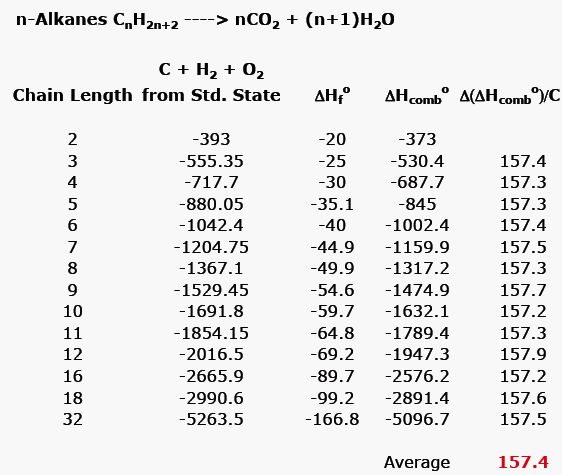
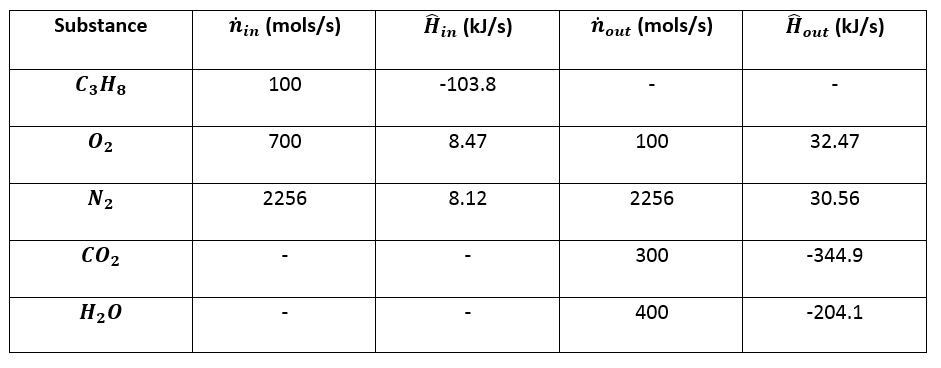
Table 5 from The Heats of Chemical Reactions: the Van't-Hoff Equation and Calorimetry | Semantic Scholar
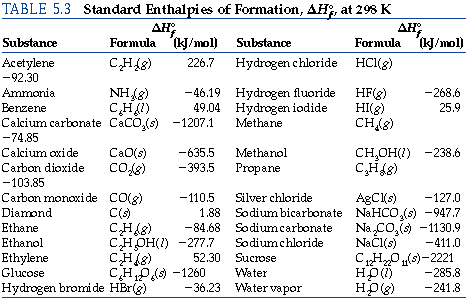
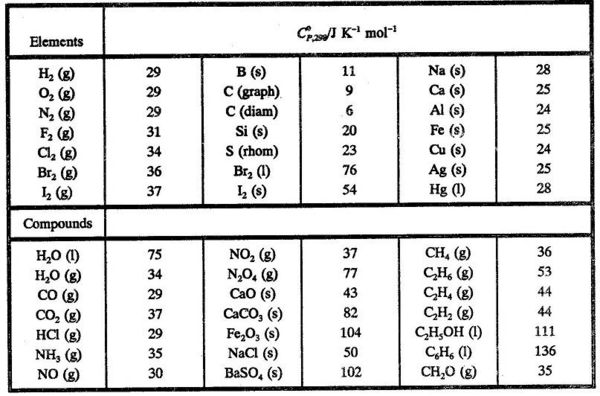
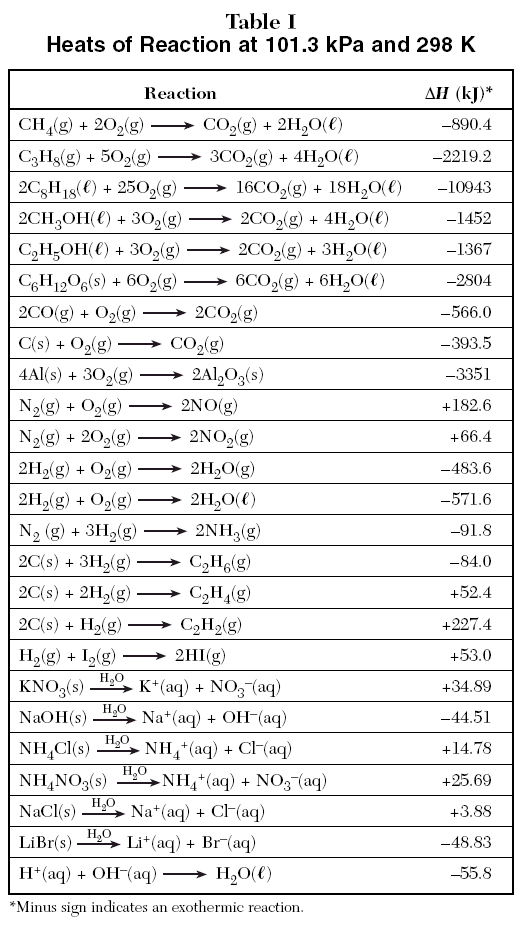
Calculate the enthalpy of reaction for the reaction "CH"_3"COOH" + "H"_2"O" -> "CH"_3"CH"_2"OH" + "O"_2? | Socratic