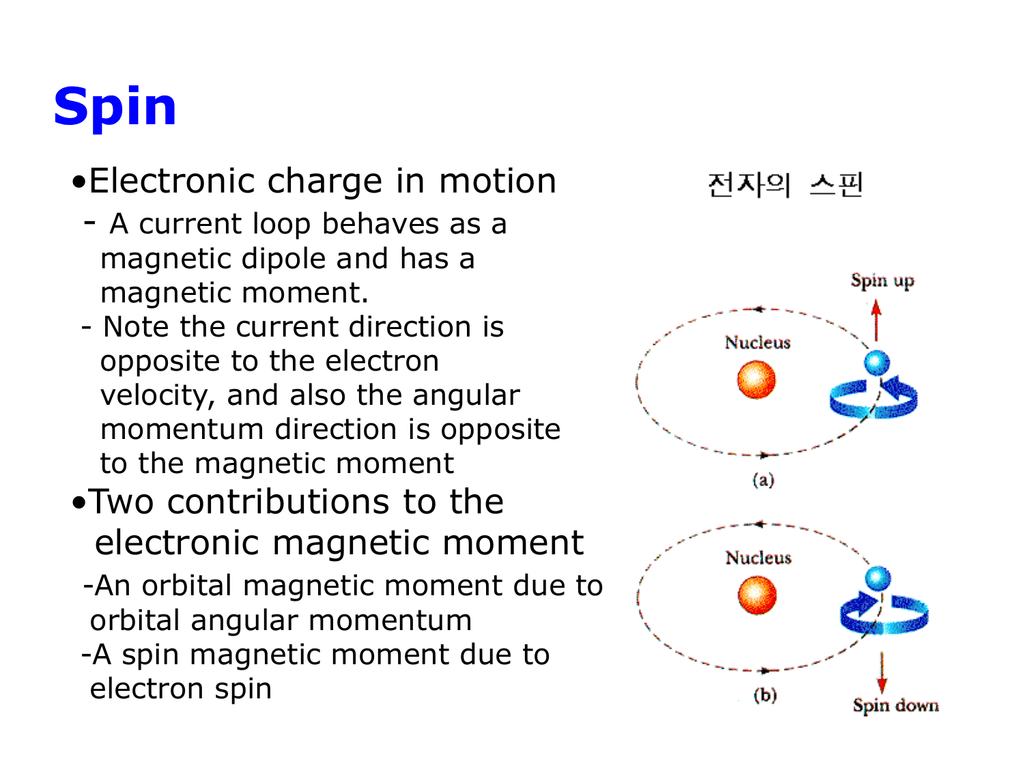
Calculate the spin only magnetic moment of La^3+. - Sarthaks eConnect | Largest Online Education Community
56.Spin only magnetic moment of Mnx+ ion is root 15B.M.Then what is tge value of X OPTIONS:A)6 B)4 C)2 D)8

The spin magnetic moment per formula unit of ZnyFe3−yO4 for y = 0 and 1. | Download Scientific Diagram
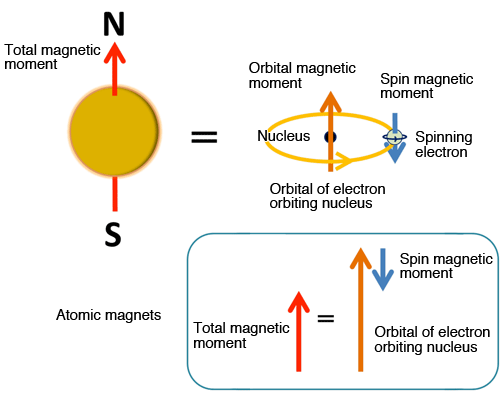
High-Accuracy Magnetic Property Measurement Method by Separating Spin and Orbital Magnetic Moments (Press Release) — SPring-8 Web Site

An electron undergoes a circular orbit, as shown in the diagram. (A) Draw the angular momentum, orbital magnetic moment and spin magnetic moment (your choice of direction) vectors on the diagram. (B)
The magnetic moment vectors µs and µ, associated with the intrinsic spin angular momentum S - Sarthaks eConnect | Largest Online Education Community


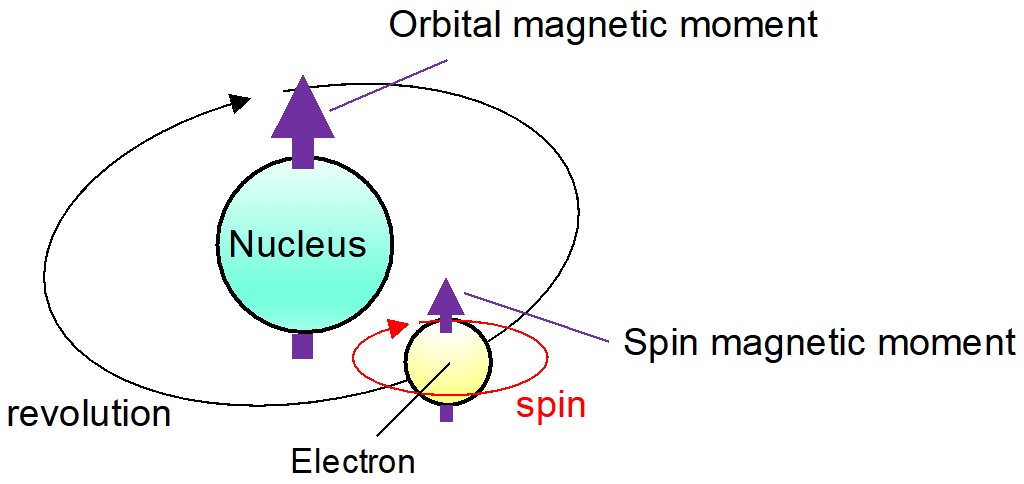
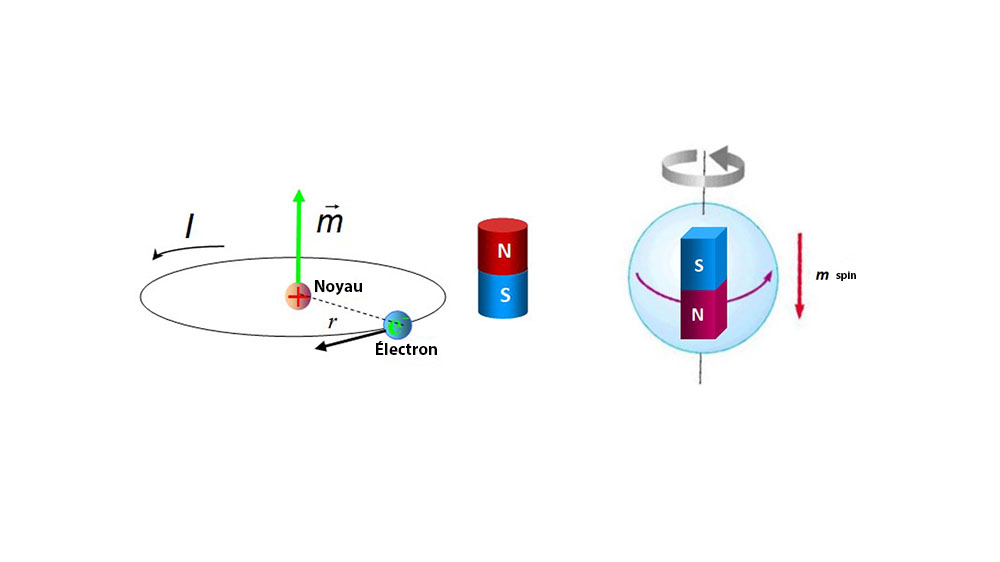
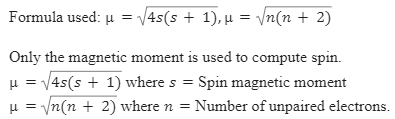
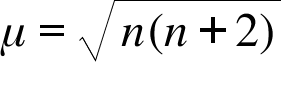

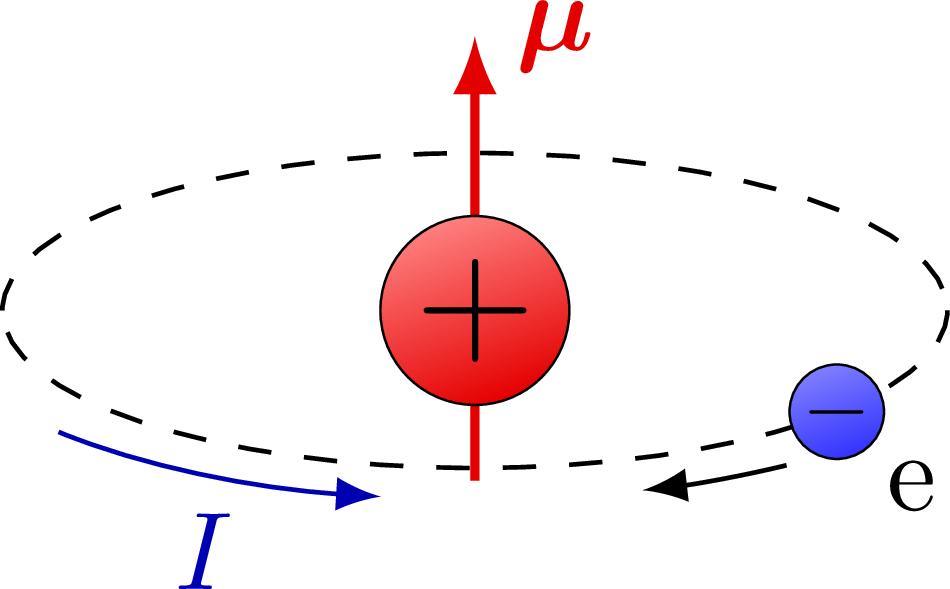
![Calculate magnetic moment of Fe^{3+} in [Fe(CN)_6]^{3-} and in [Fe(H_2O)_6]^{3-}. Calculate magnetic moment of Fe^{3+} in [Fe(CN)_6]^{3-} and in [Fe(H_2O)_6]^{3-}.](https://haygot.s3.amazonaws.com/questions/1576306_1732047_ans_78403cb9299c4e63a40902c700aaa8a9.jpg)